Unlocking the Infinite: The Chemists Crafting Molecules Atom by Atom
09/27/2023
In the intricate world of chemistry, where atoms dance and molecules weave, a revolution is underway. Chemists are now wielding their molecular tweezers with unprecedented precision, unlocking a realm of endless possibilities. Inspired by the groundbreaking CRISPR-Cas9 genome-editing technology, a new method dubbed “skeletal editing” is changing the game, promising to reshape our world molecule by molecule.

Picture this: atoms, the building blocks of all matter, rearranged at will, like pieces on a cosmic chessboard. This is the vision driving the pioneering work of chemists like Mark Levin and Richmond Sarpong. At the University of Chicago and the University of California, Berkeley, respectively, they’re spearheading efforts to rewrite the molecular code of existence.
Levin, with the passion of a poet, describes chemistry as the art of creation, where scientists sculpt the very fabric of reality. It’s a sentiment echoed by his peers worldwide, united in their quest to manipulate matter at its most fundamental level.
Traditional synthetic chemistry has long been constrained by its own limitations. Like sculptors chiseling away at marble, chemists could only modify molecules at their peripheries, unable to penetrate their core. But now, with skeletal editing, they’re delving deeper, tinkering with atoms nestled within the heart of organic compounds.
The implications are staggering. Imagine drugs tailored to fit like a key in a lock, targeting diseases with pinpoint accuracy. Today, drug discovery is a painstaking process, akin to searching for a needle in a haystack. But with skeletal editing, chemists are rewriting the rules, streamlining the journey from concept to cure.
Julia Reisenbauer and Sarah Wengryniuk are among those leading the charge, pioneering reactions that insert single atoms into molecular structures. This breakthrough sidesteps the need to start from scratch, accelerating drug development and expanding the palette of available treatments.
But the impact of skeletal editing extends far beyond medicine. In the realm of materials science, chemists are reimagining polymers without traditional constraints. Aleksandr Zhukhovitskiy and his team at the University of North Carolina at Chapel Hill have developed reactions to synthesize vinyl polymers, paving the way for sustainable alternatives to petrochemical plastics.
Imagine a world where plastics are not synonymous with pollution but are instead crafted from renewable sources, effortlessly recycled with minimal environmental impact. It’s a future within our grasp, thanks to the ingenuity of chemists embracing skeletal editing.
Yet, challenges remain. Skeletal editing is still in its infancy, with only a fraction of its potential realized. Scaling up reactions, ensuring sustainability, and navigating the complexities of molecular design pose formidable hurdles. But as Levin notes, progress is swift, with each new discovery bringing us closer to unlocking the full potential of skeletal editing.
Beyond practical applications, skeletal editing is reshaping the very essence of chemistry itself. It’s a paradigm shift, challenging chemists to dream bigger, to imagine the unimaginable. In the hands of these molecular architects, atoms are no longer mere building blocks but the raw material for a symphony of creation.
As we peer into the future, the possibilities seem boundless. Automated synthesis laboratories, personalized medicines tailored to individual genetic profiles – these are not the dreams of science fiction but the promises of skeletal editing.
In the fight against cancer, personalized precision medicine could revolutionize treatment, offering hope where once there was only despair. And in the realm of materials science, sustainable alternatives to plastics could usher in a new era of environmental stewardship.
But perhaps the true beauty of skeletal editing lies not in its practical applications but in its capacity to inspire. In the eyes of chemists like Sarpong, the true value lies in the way it challenges us to think differently, to see the world through a new lens.
In the endless dance of atoms, we find the spark of creation. And with skeletal editing, we stand on the threshold of a new frontier, where the only limit is our imagination.
Revolutionizing Cancer Screening: Detecting RNA Biomarkers with Unprecedented Precision
08/16/2023
Cancer. It’s a word that strikes fear into the hearts of millions around the world. Despite significant advancements in medical science, the battle against this relentless disease rages on. But what if there was a way to catch cancer before it even had a chance to spread? What if we could detect the presence of cancerous cells with unparalleled accuracy, long before they wreak havoc on the body?
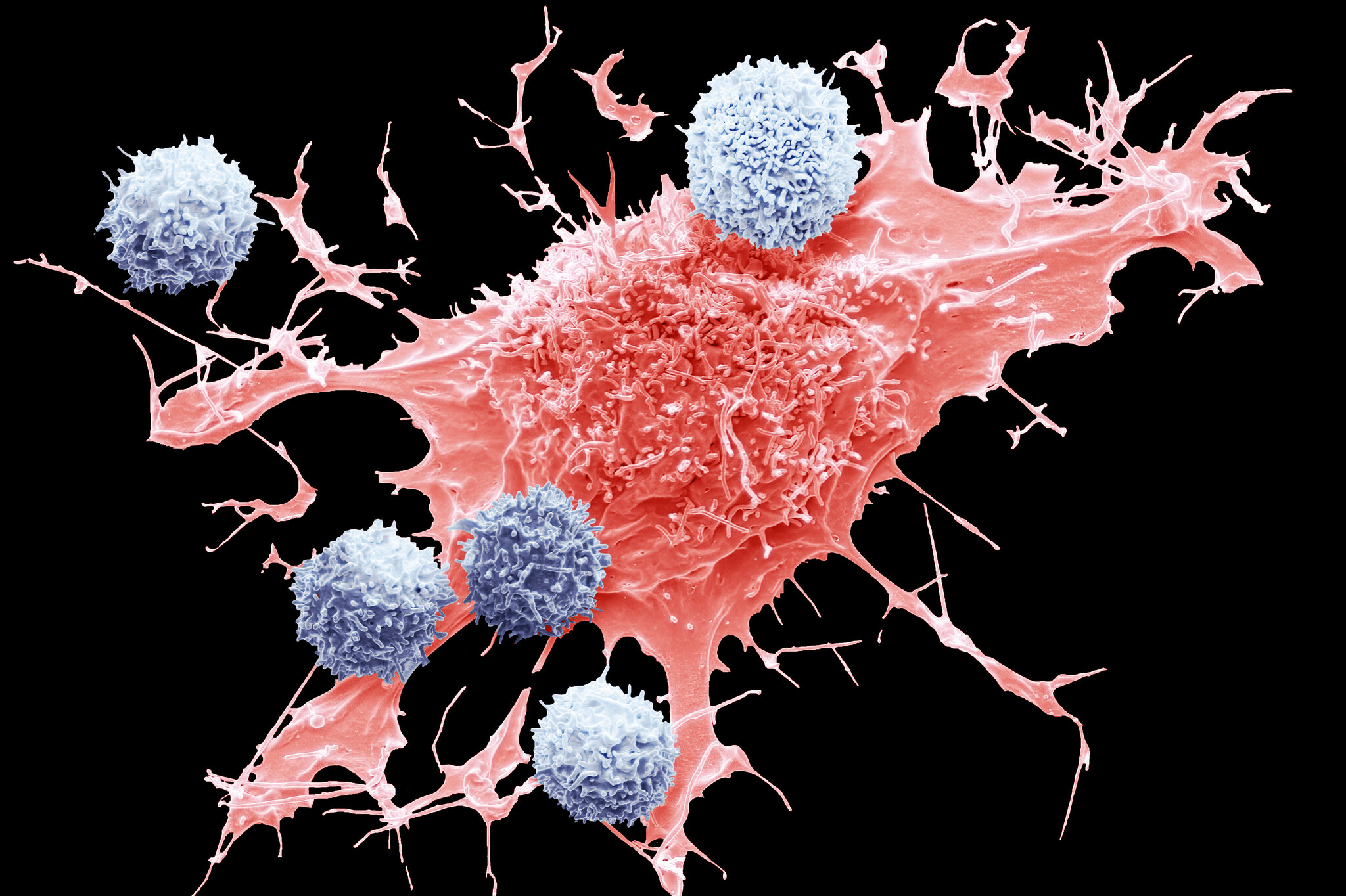
Enter the groundbreaking research on the electrical detection of RNA cancer biomarkers at the single-molecule level. Led by a team of brilliant minds, this innovative approach offers a glimmer of hope in the fight against cancer. Published in Scientific Reports, the study unveils a new frontier in early cancer detection—one that holds immense promise for improving patient outcomes and reducing mortality rates.
At the heart of this pioneering research lies the concept of liquid biopsies. Unlike traditional tissue biopsies, which can be invasive and painful, liquid biopsies offer a non-invasive alternative for detecting cancer. By analyzing biomarkers present in body fluids like blood or saliva, liquid biopsies provide a window into the body’s inner workings, offering valuable insights into the presence of cancerous cells.
However, detecting cancer biomarkers in liquid biopsies poses a significant challenge. With low concentrations and mutation frequencies, these biomarkers can be elusive, making them difficult to identify amidst the sea of genetic material present in body fluids. But where others see obstacles, these researchers see opportunities.
Harnessing the power of nanoscience and nanotechnology, the team developed a cutting-edge method for detecting RNA cancer biomarkers with unprecedented precision. Utilizing a technique known as Scanning Tunneling Microscopy (STM)-assisted break junctions, they were able to achieve single-molecule detection and identification of RNA sequences—a feat previously thought to be impossible.
So, how does it work? Imagine a nanoscale device capable of forming single-molecule junctions between two electrodes. When an RNA biomarker binds to both electrodes, it closes the biomolecular electronics circuit, resulting in a specific electrical fingerprint that can be measured. This fingerprint, known as conductance, provides vital information about the sequence and structure of the RNA molecule, allowing researchers to differentiate between cancerous and healthy cells with unparalleled accuracy.
In their study, the team focused on a well-known oncogene called KRAS, which is frequently mutated in various cancers. By designing DNA probes complementary to specific KRAS mutations, they were able to selectively target and detect cancer biomarkers with remarkable sensitivity and specificity.
What sets this approach apart is its ability to discriminate between mutant and wild-type RNA sequences, even when they differ by just a single base. This level of precision is unprecedented in the field of cancer detection, offering new hope for early diagnosis and treatment.
But perhaps the most remarkable aspect of this research is its potential to revolutionize cancer screening as we know it. With the ability to detect cancer biomarkers at the single-molecule level, liquid biopsies could become a powerful tool for identifying cancer at its earliest stages, long before symptoms manifest.
Imagine a future where routine check-ups include a simple blood test that can detect cancer with the same ease as checking cholesterol levels. Imagine a world where cancer is no longer a death sentence, but a manageable condition that can be treated with targeted therapies tailored to each individual’s unique genetic makeup.
The possibilities are endless, and the implications are profound. With further refinement and development, this groundbreaking technology could pave the way for a future where cancer is not feared, but conquered. And for millions of people around the world, that future can’t come soon enough.
Unlocking the Gut’s Secret: Toddlers’ Microbiome Could Foretell Obesity Risk
05/14/2023
In the intricate tapestry of our bodies, a lesser-known player has emerged as a potential herald of future health woes: the gut microbiome. Recent research, led by Gaël Toubon from the Université Sorbonne Paris, suggests that the bacteria inhabiting a toddler’s gut could hold clues to their future weight status. This groundbreaking study, analyzing data from 512 infants as part of a broader investigation tracking 18,000 French-born children, has illuminated a fascinating connection between early-life gut bacteria and the risk of obesity later in life.

Imagine a crystal ball that peers into a child’s future, revealing whether they’ll grapple with weight issues down the road. While it may sound like the stuff of science fiction, this study hints at a remarkable possibility: the composition of a toddler’s gut microbiome could serve as a predictive tool for obesity risk. By examining the body mass index (BMI) of participants between the ages of two and five, researchers unearthed a striking correlation between the ratio of two bacterial types—Firmicutes to Bacteroidetes—and the likelihood of obesity.
The gut, often hailed as our “second brain,” plays a pivotal role in digesting food, absorbing nutrients, and safeguarding against disease. In the early stages of life, as the microbiome undergoes dynamic changes, disruptions to its development can reverberate through adulthood, manifesting in conditions like inflammatory bowel disease, type 1 diabetes, and yes, childhood obesity.
Delving deeper into the microbial world within toddlers’ bellies, the study pinpointed specific bacterial groups associated with heightened BMI scores. Eubacterium hallii, Fusicatenibacter, and Eubacterium ventriosum were identified as potential culprits predisposing children to a higher risk of obesity. These findings were not confined to childhood; disparities in gut bacteria colonization were also observed in adults grappling with obesity, hinting at a continuum of microbial influence from cradle to adulthood.
But how do these minuscule microbes wield such significant sway over our weight? It all boils down to their role in fat absorption. Children boasting a higher ratio of Firmicutes to Bacteroidetes are inclined to absorb more calories from their food, laying the groundwork for weight gain in the years to come. In essence, these bacterial residents act as metabolic gatekeepers, shaping our propensity for obesity by regulating nutrient absorption.
Toubon underscores the critical interplay between gut bacteria composition and function, emphasizing that it’s not just about which bacteria are present, but what they’re doing. This nuanced understanding heralds a paradigm shift in our approach to combating obesity, highlighting the need to unravel the intricate dance of microbial dynamics within our bodies.
The implications of this research extend far beyond the realm of academia, signaling a potential revolution in preventive healthcare. By harnessing the predictive power of toddlers’ gut microbiomes, healthcare providers could intervene early, implementing targeted interventions to mitigate obesity risk before it spirals out of control. This proactive approach could pave the way for a healthier, more resilient generation, poised to sidestep the perils of obesity and its associated health complications.
As we unlock the secrets of the gut microbiome, a tantalizing frontier of possibilities emerges. From personalized dietary interventions to innovative probiotic therapies, the potential avenues for intervention are vast and promising. However, Toubon cautions that further research is needed to unravel the intricate web of microbial influences and pinpoint precise intervention strategies.
In the meantime, as we marvel at the profound insights gleaned from this study, one thing is abundantly clear: the tiny inhabitants of our gut wield far more influence than we ever imagined. In their delicate dance within the confines of our bodies, they may hold the key to unlocking a healthier future for generations to come.
Gut Feelings: How Microbe-Nurturing Drugs Could Revolutionize Mental Health
04/13/2023
In the fascinating realm where gut feelings meet cutting-edge science, a profound connection between our digestive system and mental well-being is being unveiled. Picture this: a bustling community of microbes within your gut, not unlike tiny residents influencing the course of your thoughts and emotions. It might sound like the plot of a whimsical comic strip, but this concept is far from fiction. In fact, it could very well shape the future of mental health treatment.
Scientists have long been intrigued by the intricate interplay between the gut and the brain, known as the gut-brain axis. This axis serves as a communication highway, where signals travel bidirectionally, influencing mood, cognition, and behavior. Recent research has uncovered the pivotal role of gut microbes in orchestrating this dialogue, offering promising avenues for novel therapeutic interventions.
Enter psychobiotics: a groundbreaking approach harnessing the power of beneficial gut bacteria to alleviate mental health disorders such as anxiety and depression. Imagine a world where instead of relying solely on traditional medication or therapy, we could manipulate our microbial communities to support our mental well-being. It may sound revolutionary, but the science behind it is compelling.
Studies have demonstrated that altering the composition of gut microbes, either by introducing specific strains or nurturing existing ones, can yield profound effects on mental health. For instance, experiments with germ-free mice have shown that inoculating them with “good” bacteria leads to reductions in stress, anxiety, and depression-like behaviors. These findings highlight the potential of psychobiotics as a novel therapeutic approach
But the journey from lab bench to bedside is not without its complexities. One major challenge lies in understanding the intricate relationship between gut microbes and the brain. Researchers are still deciphering which microbial strains influence specific mental health disorders and whether it’s the microbes themselves or the chemicals they produce that exert therapeutic effects
Moreover, individual variability in gut microbiomes adds another layer of complexity. Factors such as diet, inflammation, and genetics contribute to unique microbial compositions, emphasizing the need for personalized treatment approaches. Despite these challenges, the potential of psychobiotics to revolutionize mental health care cannot be overstated.
Excitingly, research is already yielding promising results. Studies have shown that dietary interventions aimed at promoting the growth of beneficial gut bacteria can lead to improvements in perceived stress and cognitive function. From fiber-rich foods to fermented delights like kimchi and kefir, the key to nurturing a healthy microbiome may lie in the foods we consume.
Additionally, prebiotics – substances that feed beneficial gut bacteria – have shown promise in enhancing problem-solving abilities in individuals with psychosis. These findings underscore the profound impact of diet on mental health and highlight the potential of dietary interventions as adjunct therapies for psychiatric conditions.
Looking ahead, the future of mental health treatment may very well be shaped by our understanding of the gut-brain axis. As researchers delve deeper into the mechanisms underlying this intricate relationship, the potential for novel therapeutic interventions continues to expand. From personalized psychobiotic regimens to dietary interventions tailored to individual microbiome profiles, the possibilities are endless.
In conclusion, the emerging field of psychobiotics offers a glimpse into a future where mental health care is as much about nurturing our gut microbes as it is about addressing psychological symptoms. As we unravel the mysteries of the gut-brain axis, we may find that the key to mental well-being lies within the trillions of microorganisms residing within us. So, the next time you have a gut feeling, remember: it might just be your microbes speaking to you.
Is Space the Next Frontier for Medicine?
03/10/2023
In the quest to unravel the mysteries of medicine and push the boundaries of scientific discovery, humanity has often looked beyond the confines of our planet. As we stand at the precipice of a new era in healthcare, one question looms large: does the future of medicine lie in space?

For decades, space has captured our imagination as the realm of astronauts and cosmic exploration. However, in recent years, a new frontier has emerged – one where the vast expanse of the cosmos holds the promise of groundbreaking medical advancements. At the forefront of this revolution is a cadre of pioneering scientists, entrepreneurs, and visionaries who are leveraging the unique environment of space to unlock the secrets of human health.
At the heart of this endeavor is the International Space Station (ISS), a floating laboratory orbiting 250 miles above the Earth. Here, researchers are conducting experiments free from the constraints of gravity, offering unparalleled insights into the inner workings of biological processes. Israeli entrepreneur Yossi Yamin, CEO of SpacePharma, is one such trailblazer, spearheading efforts to miniaturize experiments and send them into orbit aboard SpaceX rockets. These experiments, ranging from the behavior of leukemia cells to the production of lab-grown steak, have yielded transformative insights with far-reaching implications for medicine.
The absence of gravity in space unveils a realm where traditional barriers to medical research are dissolved. On Earth, gravity distorts cell communication and hampers the growth of protein crystals essential for understanding diseases like cancer and viruses. However, in the weightlessness of space, scientists can observe these processes in their purest form, paving the way for novel therapeutic interventions. Prof. Thais Russomano, a leading expert in space medicine, emphasizes the importance of studying protein structures in space, which offer unparalleled clarity and precision compared to their Earth-grown counterparts.
The impact of space-based research on medicine has already been profound. Massachusetts-based biotech company MicroQuin credits experiments conducted on the ISS for kickstarting a new pipeline of drugs targeting ovarian and breast cancers, traumatic brain injury, Parkinson’s, and influenza. By crystallizing proteins in space, MicroQuin has unlocked new avenues for drug development, harnessing the power of zero gravity to tackle some of humanity’s most pressing health challenges.
Yet, the journey to space is not without its challenges. The exorbitant cost of space travel, estimated at $7.5 million per experiment, poses a significant barrier to entry for many researchers. However, the rise of private space companies like Axiom Space presents a glimmer of hope. Axiom Space aims to replace the ISS with a commercial space station, funded in part by space tourism revenue, offering unprecedented opportunities for scientific exploration.
Moreover, advancements in automation and technology are poised to revolutionize space-based research, making it more accessible and cost-effective than ever before. Companies like Ice Cubes are streamlining the process of conducting experiments in low Earth orbit, eliminating the need for a dedicated space station and democratizing access to the cosmos.
As we peer into the future, the implications of space medicine are profound and far-reaching. Stem cell research conducted in space holds the promise of regenerative therapies capable of repairing damaged organs and revolutionizing transplant medicine. The prospect of growing organs in space, free from the constraints of gravity, tantalizes with the possibility of a future where organ shortages are a thing of the past.
In the grand tapestry of human endeavor, the convergence of space exploration and medicine represents a watershed moment in history. As we venture into the unknown depths of space, we are not only exploring the cosmos but also unlocking the secrets of human health and paving the way for a future where disease and suffering are relics of the past. The future of medicine may indeed lie in space, where the final frontier beckons with the promise of hope, discovery, and healing.
“Aptamer: Revolutionizing Cancer Treatment from the Heart of York’s Biotech Scene”
01/09/2023
In the heart of the bustling biotech scene in York, a small yet mighty company is quietly revolutionizing the landscape of cancer treatment. Aptamer Group, nestled next to the University of York, is making waves with its groundbreaking work in precision medicine, particularly in the realm of cancer treatment

At the forefront of their innovation is the development of aptamers – synthetic antibodies crafted from fragments of DNA. These aptamers, akin to molecular keys, possess the remarkable ability to bind to specific targets within the body, such as tumor cells. In a world where cancer treatment often comes with harsh side effects and limited efficacy, the promise of precision medicine holds immense potential for transforming patient outcomes.
Dr. David Bunka, the company’s chief technical officer, paints a vivid picture of their work as he guides visitors through their state-of-the-art lab. Here, advanced robotics tirelessly screen millions of molecules in a fraction of the time it would take traditional methods. The result? A streamlined process that accelerates the development of aptamers for a range of medical applications.
But Aptamer Group’s impact extends far beyond the confines of their lab. With partnerships spanning the globe, including collaborations with pharmaceutical giants like AstraZeneca and Takeda, their reach is truly global. Together, they are exploring new frontiers in cancer treatment, from targeted therapies to diagnostic tools that promise earlier detection and more effective interventions.
One of the key advantages of aptamers lies in their synthetic nature. Unlike traditional antibodies, which are often sourced from living beings and can pose ethical and logistical challenges, aptamers are created using cutting-edge DNA technology. This not only expedites the production process but also offers greater stability and versatility in application.
Nick Turner, a professor in bioanalytical chemistry, underscores the ethical and practical advantages of aptamers. With no reliance on animal models and enhanced stability, these synthetic antibodies offer a beacon of hope for both patients and researchers alike.
Moreover, the speed at which Aptamer Group operates is nothing short of impressive. With the ability to develop aptamers in as little as 15 days, compared to the months-long timeline for traditional antibodies, they are setting a new standard for efficiency in the biotech industry. This agility is crucial in the fight against cancer, where every moment counts in the race to save lives.
But the journey hasn’t been without its challenges. Aptamer Group, like many biotech startups, has faced hurdles in recruitment, exacerbated by factors such as the post-Brexit hiring landscape and a lack of affordable housing in York. Yet, despite these obstacles, their commitment to pushing the boundaries of science remains unwavering.
Their recent collaboration with Deepverge on Covid-19 detection in wastewater highlights the versatility of their technology. Beyond cancer treatment, Aptamer Group’s aptamers hold promise in addressing a myriad of medical challenges, from infectious diseases to environmental monitoring.
As the global aptamer market continues to soar, projected to reach billions in annual revenue by 2030, Aptamer Group stands poised at the forefront of this burgeoning industry. Their journey from a humble basement startup to a trailblazing biotech firm is a testament to the power of innovation and perseverance.
In the ever-evolving landscape of cancer treatment, Aptamer Group shines as a beacon of hope, forging new paths towards more effective, personalized therapies. With each breakthrough, they inch closer to their vision of a world where cancer is not just treatable, but beatable. And in the bustling biotech scene of York, their legacy continues to unfold, one aptamer at a time.
